|
Color is generally defined as the
characteristic of any object that’s described
in terms of hue, lightness, and saturation. In 1666
Sir Isaac Newton, (Mr. Gravity) through experiments
with a prism, laid a scientific foundation for understanding
color. Newton showed that a prism could break up
white light into a range of colors, which he called
the spectrum.
|

Figure 1.1 - Visible Spectrum |
Newton noted that the spectrum
was continuous, but decided to use seven color names
(red,
orange, yellow, green, blue, indigo, and violet) by analogy
with the seven notes of the musical scale. (Britannica: "color") Although
Newton stated that there were seven colors in the spectrum,
he
realized that colors other than those in the spectral
sequence do exist, but noted that:
All the colors
in the universe which are made by light, and depend
not on the power of imagination, are either the colors
of homogeneal lights (i.e., spectral colors), or
compounds
of these. (Britannica: "color")
There are three categories for color types. First, primary
colors consist of red, blue, and yellow. Combinations
(or
as Newton put it “compounds”) of these fall
into the other two categories: secondary colors and tertiary
colors (Color
Harmony 16).
Britannica Online notes that there are three attributes
that sufficiently distinguish one color from all other
perceived colors. First, the hue is that aspect of color
usually associated with terms such as red, orange, yellow,
and so on. Second, saturation (also known as chroma, or
tone) refers to relative purity. When a pure, vivid,
strong
shade of blue is mixed with a variable amount of white,
weaker or paler blues are produced, each having the
same
hue but a different saturation. Lastly, light of any given
combination of hue and saturation can have a variable
brightness
or intensity, which is dependent on the level of energy
present (Britannica: "color").
Chromatic, nonchromatic,
and achromatic colors are visible to the human eye. Chromatic
colors are the ones defined by Newton (e.g. red, indigo,
yellow). Examples of nonchromatic colors are brown, pink,
and magenta. Achromatic colors are applied to black, grey,
and white. Britannica Online states that according to
some
reports, humans can distinguish some 10 million colors,
all of which derive from two types of light mixture: additive
and subtractive. Additive mixture involves the addition
of spectral components and subtractive mixture concerns
the subtraction (or absorption) of parts of the spectrum
(Britannica: "color").
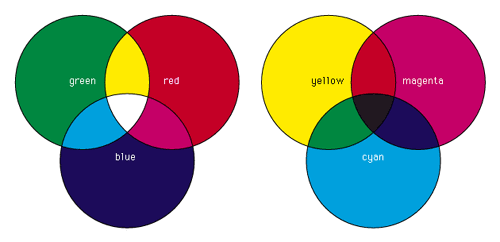
Figure
1.6 - color Mixture
The three additive colors are red, green, and blue. By
additively mixing these colors in varying amounts
almost
all other colors can be produced. Moreover, when the three
primary colors are mixed together in equal amounts
white
is produced.
Subtractive color mixing
involves the absorption and selective transmission or
reflection
of light. This usually happens when mixing colorants like
pigments or dyes or when colored filters are used to
cover
a beam of light (Britannica: "color").
About Light
 |
In the field of physics,
color is associated specifically with electromagnetic
radiation
of a range of wavelengths visible to the human eye. The
radiation of these wavelengths comprises that portion
of
the electromagnetic spectrum also known as the visible
spectrum (i.e. light) (Britannica: "color").
Light, a small piece of the electromagnetic spectrum is
the only visible form of electromagnetic radiation. Light
has common characteristics with both waves and particles.
It can be thought of as a stream of minute energy packets
radiated at varying frequencies in a wave motion (Britannica: "color").
A wavelength (the distance between corresponding points
of two consecutive waves) is often expressed in units
of
nanometers (or 1 nm = 10^-9 meters). The wavelengths that
make up visible light range from about 400 nm at the violet
end of the spectrum to 700 nm at the red end. In an interesting
side note, the limits of the visible spectrum are not
exact
for the human race. All humans have different exact upper
and lower limits within the spectrum. As wavelengths get
shorter the spectrum extends to include ultraviolet and
continues through X-rays, gamma rays, and cosmic rays.
As
wavelengths get longer infrared rays (which can be felt
as heat), microwaves, and radio waves are included in
the
spectrum (Britannica: "color").
The Measurement
of Color
The measurement of color
is known as colorimetry. It is difficult to describe the
color of a specific spectral energy distribution because
the eye perceives only a single color for any given energy
distribution. So, to measure color it is necessary to
express color measurements using a perception-related
method. One
method is called the tristimulus system. This system is
based on visually matching a color under standardized
conditions
against the three primary colors (red, yellow, and blue).
The three results (called tristimulus values) are expressed
as X, Y, and Z respectively. Such data can be graphically
represented on a chromaticity diagram (see figure 1.7)
(Britannica: "color").
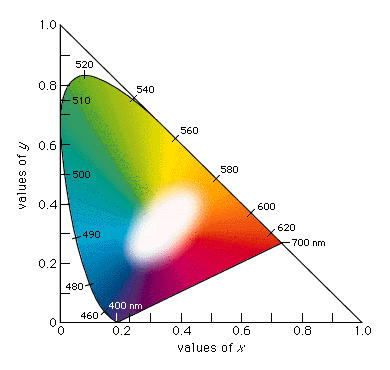
Figure
1.7 - Chromaticity Diagram
This diagram is based on the values of x, y, and z (where
x = X/(X+Y+Z), y = Y/(X+Y+Z), and z = Z/(X+Y+Z)). Furthermore,
because x + y + z = 1, if two values are known, the third
can always be calculated and the z value is usually
omitted.
The x and y values together constitute the chromaticity
of a sample (Britannica: "color").
|